How To Draw Atomic Orbitals
How To Draw Atomic Orbitals - This video goes over how to properly draw orbital diagrams for an element, after determining the electron configuration. Calculate bond orders based on molecular electron configurations. Web © 2024 google llc. Web atomic orbitals are mathematical functions that describe the wave nature of electrons (or electron pairs) in an atom. Web the electrons in an atom are arranged in shells that surround the nucleus, with each successive shell being farther from the nucleus. O2 = (8 − 4) 2 = 2 o 2 = ( 8 − 4) 2 = 2. 366k views 10 years ago chemistry tutorials: Subshells are designated by the letters s , p , d , and f , and each letter indicates a different shape. Web bohr's atomic model and its limitations (de broglie's equation, heisenberg’s uncertainty principle), concept of shells, subshells, orbitals. They offer a way to calculate the probability of finding an electron in a specified region around the nucleus of the atom. Electron shells consist of one or more subshells, and subshells consist of one or more atomic orbitals. So the atomic orbital diagram is simply those orbitals in that order of energy. This video goes over how to properly draw orbital diagrams for an element, after determining the electron configuration. Web this tutorial is about orbitals. Quantum numbers, shapes of s,. Orbital diagrams are a visual way to show where the electrons are located within an atom. Each box represents one orbital, and each arrow indicates one electron. This knowledge is a precursor to chemical bonding. All orbitals with values of n > 1 and ell = 0 contain one or more nodes. 1s 2 2s 2 2p 1. Web this tutorial is about orbitals. Web so, how do these mathematically defined orbitals fit in with the electron shells we saw in the bohr model? With oxygen, you know that the atomic orbital potential energies go in the following order: Keep in mind the energy of the atomic orbitals and molecular orbitals! The use of quantum theory provides the. Web the electrons in an atom are arranged in shells that surround the nucleus, with each successive shell being farther from the nucleus. We can break each electron shell down into one or more subshells, which are simply sets of one or more orbitals. Get a 10 bullets summary of the topic. Web in orbitals diagrams, the orbitals are shown. Web how might one draw atomic and molecular orbital diagrams? By the end of this section, you will be able to: Orbital diagrams are a visual way to show where the electrons are located within an atom. Web this tutorial is about orbitals. For example, the orbital diagram of li can be shown as: This knowledge is a precursor to chemical bonding. The shape of s orbitals. Web © 2024 google llc. We can break each electron shell down into one or more subshells, which are simply sets of one or more orbitals. I will use oxygen ( o2(g)) as an example. For example, the orbital diagram of li can be shown as: O2 = (8 − 4) 2 = 2 o 2 = ( 8 − 4) 2 = 2. Web the four chemically important types of atomic orbital correspond to values of ℓ = 0, 1, 2, and 3. Orbitals with ℓ = 0 are s orbitals and are spherically. With oxygen, you know that the atomic orbital potential energies go in the following order: Web fill molecular orbitals using energy and bonding properties of the overlapping atomic orbitals. We calculate the bond order as. Get a 10 bullets summary of the topic. Web this tutorial is about orbitals. I will use oxygen ( o2(g)) as an example. All orbitals with values of n > 1 and ell = 0 contain one or more nodes. Orbitals with ℓ = 0 are s orbitals and are spherically symmetrical, with the greatest probability of finding the electron occurring at the nucleus. They offer a way to calculate the probability of finding. With oxygen, you know that the atomic orbital potential energies go in the following order: 1s 2 2s 2 2p 1. Web atomic orbitals are mathematical functions that describe the wave nature of electrons (or electron pairs) in an atom. Web in orbitals diagrams, the orbitals are shown as boxes, and the electrons in them as arrows pointing up or. Web for a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus. Electron configurations are expressed through a notation that looks like this: So the atomic orbital diagram is simply those orbitals in that order of energy. The following factors contribute to the position of one mo with respect to other mos. With oxygen, you know that the atomic orbital potential energies go in the following order: The molecular orbital energy diagram for o 2 predicts two unpaired electrons. This video goes over how to properly draw orbital diagrams for an element, after determining the electron configuration. Each box represents one orbital, and each arrow indicates one electron. Orbitals with ℓ = 0 are s orbitals and are spherically symmetrical, with the greatest probability of finding the electron occurring at the nucleus. This knowledge is a precursor to chemical bonding. O2 = (8 − 4) 2 = 2 o 2 = ( 8 − 4) 2 = 2. Web how might one draw atomic and molecular orbital diagrams? 1.9m views 3 years ago new ap & general chemistry video playlist. Web the electrons in an atom are arranged in shells that surround the nucleus, with each successive shell being farther from the nucleus. Subshells are designated by the letters s , p , d , and f , and each letter indicates a different shape. Get a 10 bullets summary of the topic.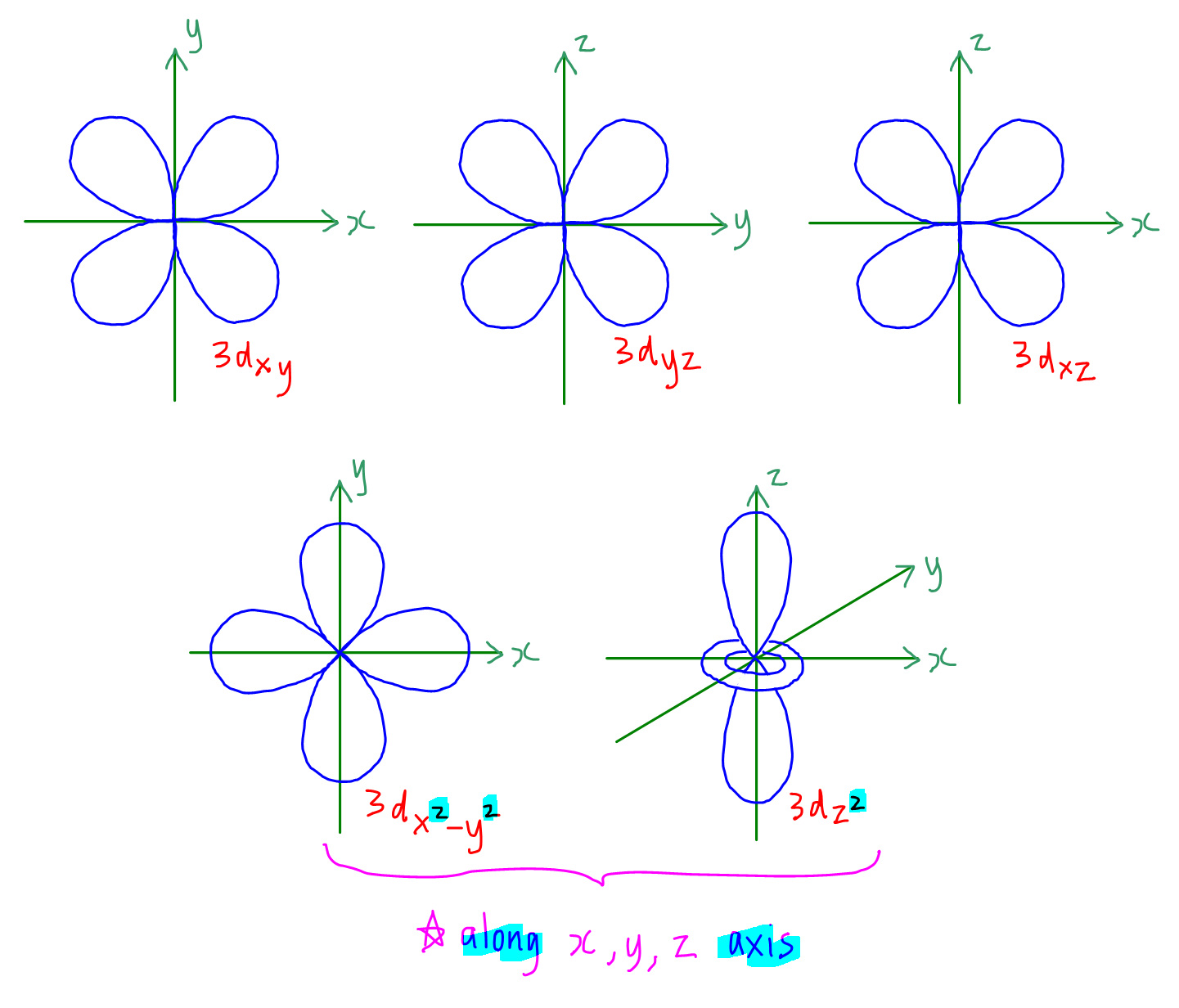
How to Draw Shapes of Orbitals

How To Draw Orbitals Deepcontrol3

Shapes of Atomic Orbitals — Overview & Examples Expii

Draw The Molecular Orbital Diagram For The Formation Class 11 Chemistry
8.3 Development of Quantum Theory CHEM 1114 Introduction to Chemistry
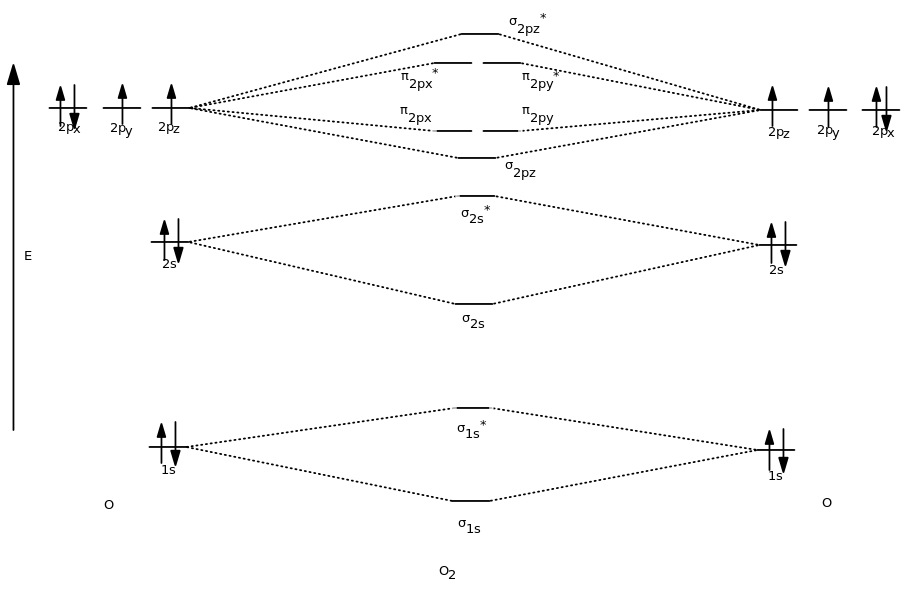
Drawing Atomic and Molecular Orbitals Diagrams for Molecules Organic

Different Types Of Orbitals
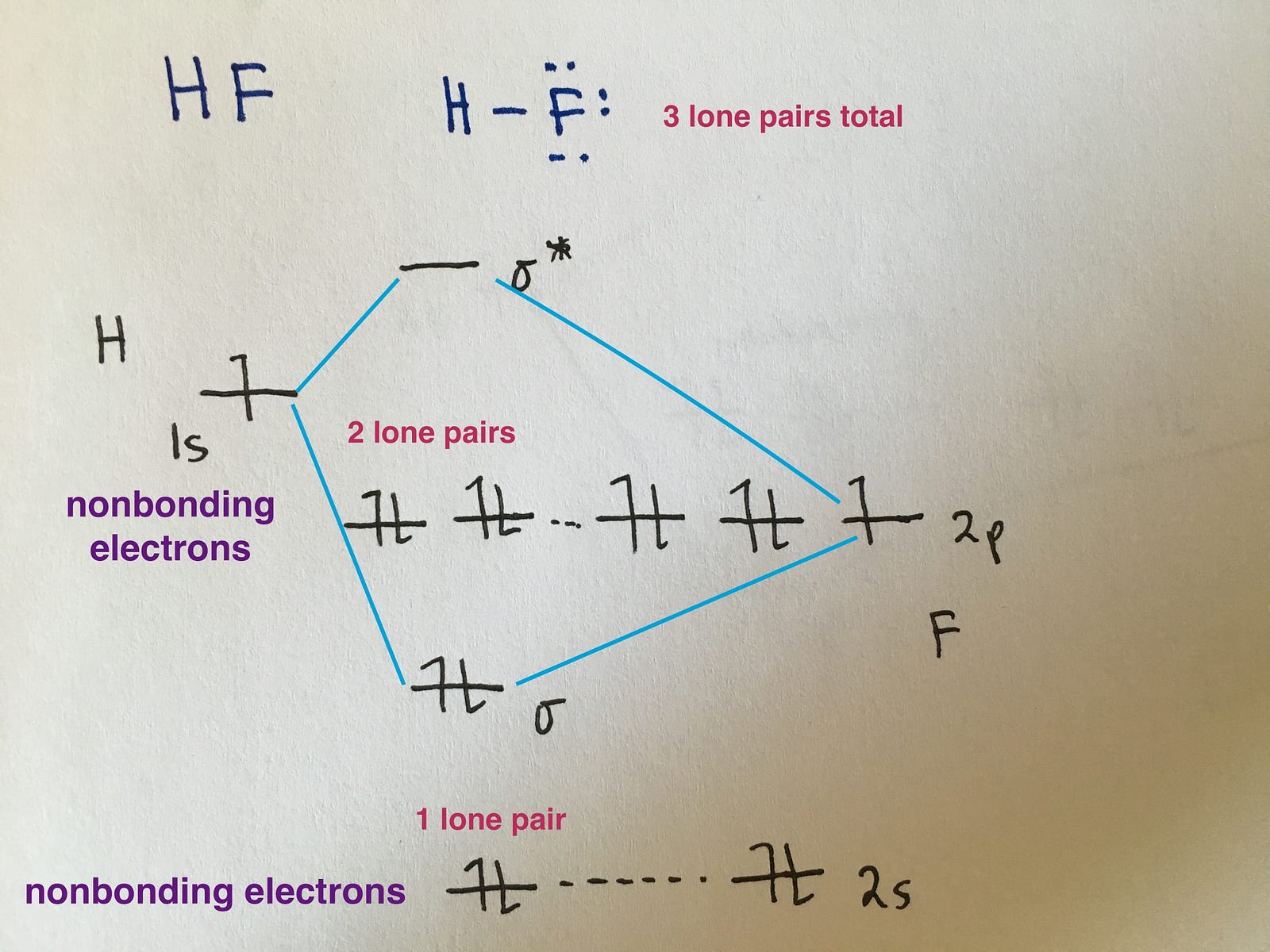
Molecular Orbital Diagrams simplified Megan Lim Medium
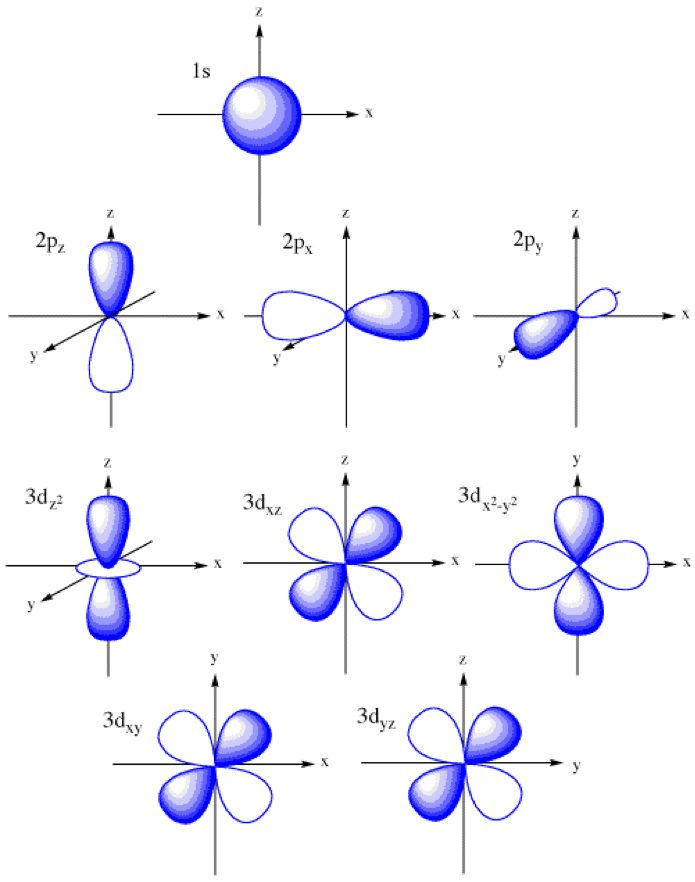
Atomic orbitals explained polizhuge

How To Draw Orbitals Deepcontrol3
Web On A Tight Schedule?
We Can Break Each Electron Shell Down Into One Or More Subshells, Which Are Simply Sets Of One Or More Orbitals.
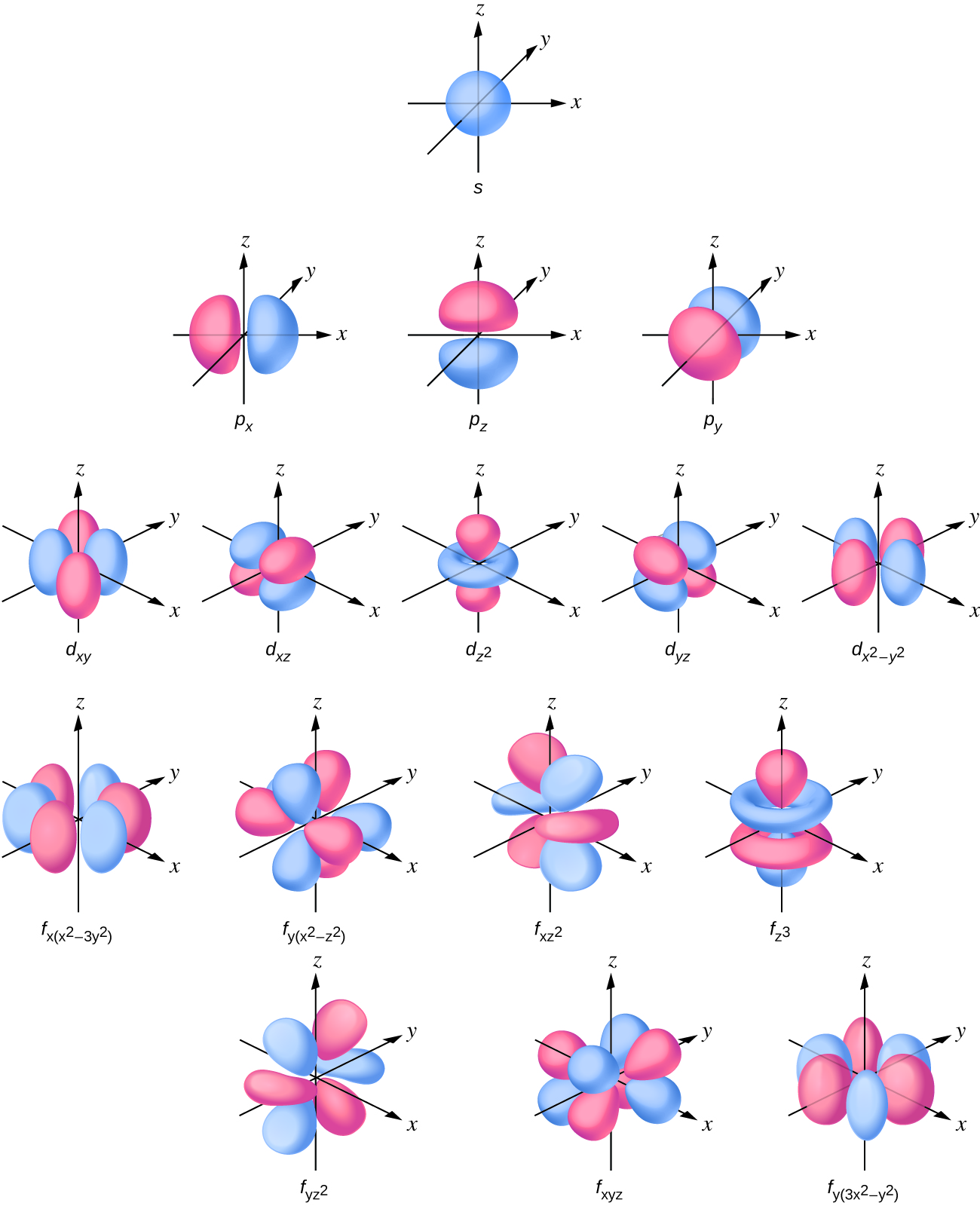
Quantum Numbers, Shapes Of S, P And D Orbitals.
You Will Also Learn How To Use Hund'.
Related Post: