Draw The Electron Configuration For A Neutral Atom Of Beryllium
Draw The Electron Configuration For A Neutral Atom Of Beryllium - Here, the electron configuration of beryllium ion (be 2+) is 1s 2. Web atomic structure and electron configuration. Draw a small circle and write the symbol in the centre. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its. The electron configuration for the be 2+ is 1s 2. In order to write the be electron configuration we first need to k. Therefore, the number of electrons in neutral atom of beryllium is 4. Web therefore, beryllium is a cation element. The shorthand electron configuration for beryllium is [he] 2s 2. Electron configuration of beryllium (be) [he] 2s 2: This electron configuration shows that the beryllium ion (be 2+) acquired the electron configuration of helium and it achieves a stable electron configuration. Using s p d f notation, what is the electron configuration for a neutral atom of beryllium? Since 1s can only hold two electrons the remaining 2 electrons for be go in the 2s orbital. A. And for the excited state, it is 1s 2 2s 1 2p 1. Its chemical symbol is be. The electronic configuration of be is [he]2s2. The total number of electrons matches the. Web for hydrogen, therefore, the single electron is placed in the 1 s orbital, which is the orbital lowest in energy (figure 6.8.1 6.8. The total number of electrons matches the. Therefore the be electron configuration will be 1s 2 2s 2. Since 1s can only hold two electrons the remaining 2 electrons for be go in the 2s orbital. Fill these 8 electrons in the following order: The electron configuration for the be 2+ is 1s 2. Energy 1 l х 5 ? Fill these 8 electrons in the following order: The full configuration for be, z = 4, is simply 1s22s2. The number of valence electrons available for beryllium atoms is 2. Electron configuration of helium (he) 1s 2: The lewis symbol of carbon depicts a “c’ surrounded by 4 valence electrons because carbon has an electron configuration of 1s 2 2s 2 2p 2. Draw a small circle and write the symbol in the centre. 1s, 2s, and then 2p. As an example, a neutral atom of gold (au) contains 79 protons in its nucleus and 79 electrons.. 1s, 2s, and then 2p. There are 2 steps to solve this one. This means that the first energy level (shell) has 1s², indicating two electrons in the 1s subshell. Draw the electron configuration for a neutral atom of beryllium. Science > chemistry library >. Fill these 8 electrons in the following order: Draw a small circle and write the symbol in the centre. As an example, a neutral atom of gold (au) contains 79 protons in its nucleus and 79 electrons. Web to find the electron configuration of oxygen: In writing the electron configuration for beryllium the first two electrons will go in the. Using s p d f notation, what is the electron configuration for a neutral atom of beryllium? Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. To begin solving this, recall that the aufbau principle guides the process of adding electrons to an atom, filling up orbitals in an order. Draw a small circle and write the symbol in the centre. This electron configuration shows that the beryllium ion (be 2+) acquired the electron configuration of helium and it achieves a stable electron configuration. Web notice that the outermost level has only one electron. Web therefore, beryllium is a cation element. Write the complete electron configuration of oxygen: This electron configuration shows that the beryllium ion (be 2+) acquired the electron configuration of helium and it achieves a stable electron configuration. Web to find the electron configuration of oxygen: To begin solving this, recall that the aufbau principle guides the process of adding electrons to an atom, filling up orbitals in an order of ascending energy levels. This. Web shorthand electron configuration full electron configuration electron shell arrangement; This means that the first energy level (shell) has 1s², indicating two electrons in the 1s subshell. Since 1s can only hold two electrons the remaining 2 electrons for be go in the 2s orbital. As an example, a neutral atom of gold (au) contains 79 protons in its nucleus and 79 electrons. The number of the principal quantum shell, n, the letter that designates the orbital type (the subshell, l), and Web atomic structure and electron configuration. There are 2 steps to solve this one. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its. We place one electron in the orbital that is. O electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of phosphorus. There are 2 steps to solve this one. The total number of electrons matches the. Web for a neutral atom of beryllium (be), which has an atomic number of 4, the electron configuration is written as 1s²2s². Electron configuration of beryllium (be) [he] 2s 2: Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. We describe an electron configuration with a symbol that contains three pieces of information (figure 6.25):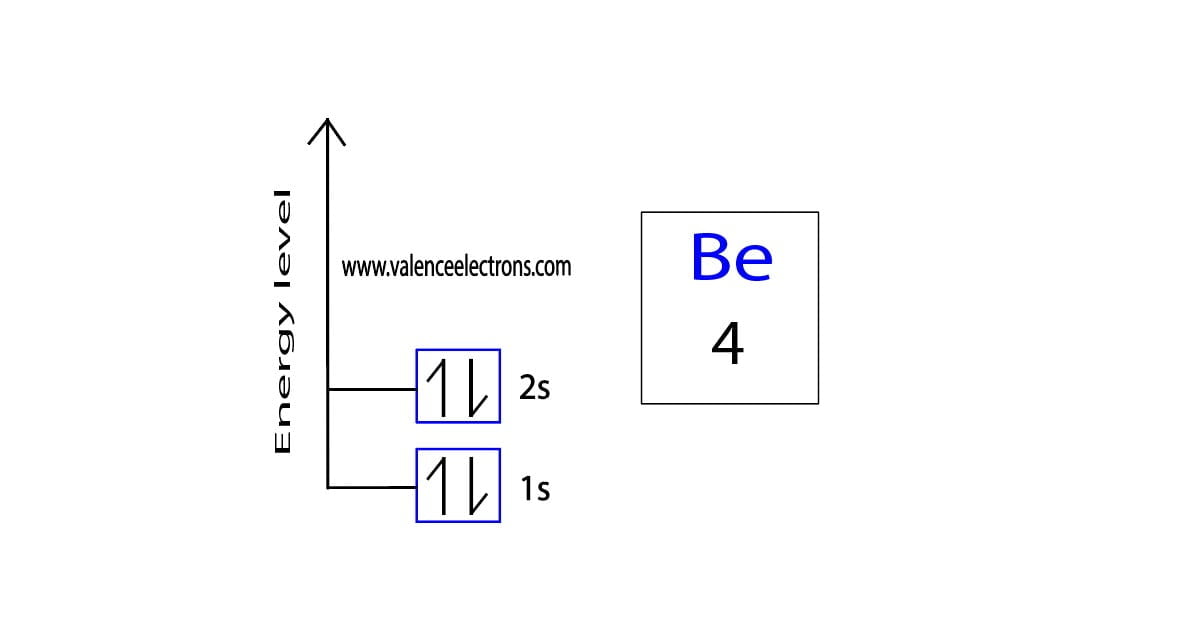
Orbital Diagram for Beryllium and Process of Drawing It
Solved Draw the electron configuration for a neutral atom of

How Can We Find A Beryllium Electron Configuration (Be)

Be 2+ Electron Configuration (Beryllium Ion) YouTube
SOLVED Draw the electron configuration for a neutral atom of beryllium.
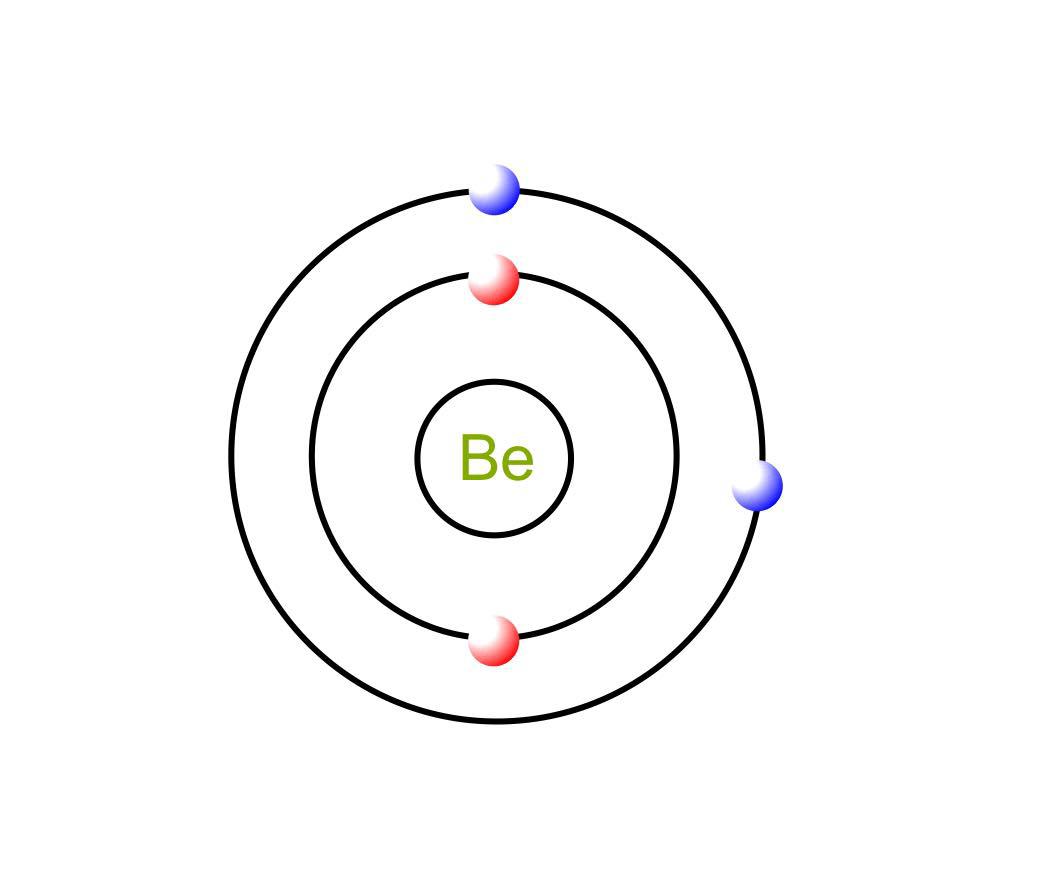
Draw a neutral Beryllium atom showing its electron shells. Quizlet
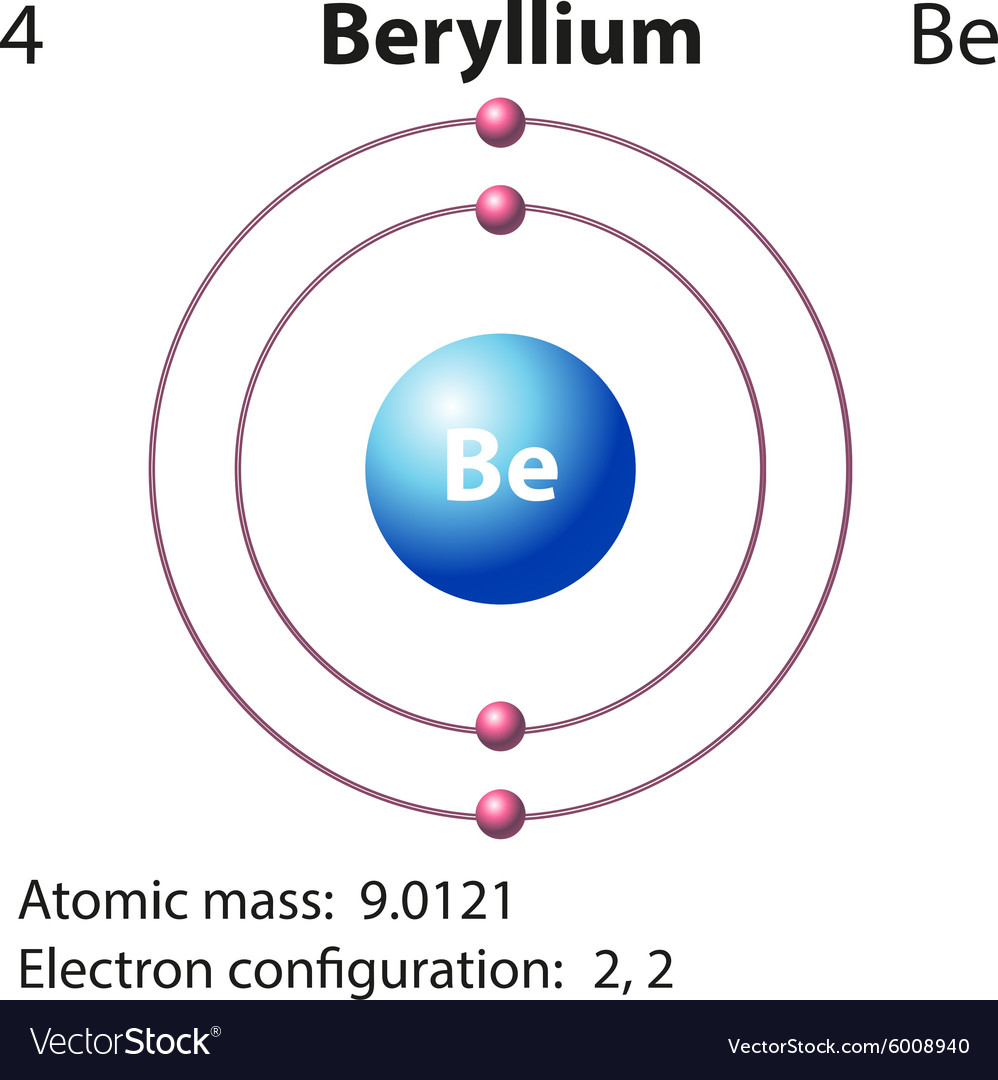
Diagram representation element beryllium Vector Image

Beryllium Electronic Configuration By SCIENCE THINK YouTube

Electronic configuration of Beryllium spdf Trick Chemistry

Beryllium(Be) electron configuration and orbital diagram
Write The Complete Electron Configuration Of Oxygen:
A Neutral Helium Atom, With An Atomic Number Of 2 ( Z = 2), Has Two Electrons.
For Early Elements, Clearly The Given Configuration Using The Noble Gas Core Does Not Save You A Lot Of Work.
Therefore The Be Electron Configuration Will Be 1S 2 2S 2.
Related Post: