Draw An Appropriate Lewis Structure For If5
Draw An Appropriate Lewis Structure For If5 - #3 indicate formal charges on the atoms, if necessary. Which has the larger bond angle? Web the following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web i quickly take you through how to draw the lewis structure of if5 (iodine pentafluoride). Write lewis symbols for neutral atoms and ions. Draw lewis structures depicting the bonding in simple molecules. We draw lewis structures to predict: For the compounds (i) if5 and (ii) ch3sh, complete the following: In short, these are the steps you need to follow for drawing a lewis structure: The iodine atom has 1 lone pair while all the five fluorine atoms have 3 lone pairs. Web thus the lewis structure of any compound can be formed using these simple steps! The if 5 lewis structure you'll need to put more than eight valence electrons on the iodine atom. For each compound draw an appropriate lewis structure, determine the molecular geometry using vsepr theory, determine whether the molecule is polar and identify the hybridization of all. The hybridization of if5 is sp3d2. This gives it ax5e1 shape. Which has the larger bond angle? Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Draw the lewis dot structure of the molecule if5 and determine the electron and molecular geometries around the i atom. Web thus the lewis structure of any compound can be formed using these simple steps! First, determine the total number of valence. This gives it ax5e1 shape. * hydrogen atoms are always terminal (only one bond) * put more electronegative elements in terminal positions. Put one electron pair in each bond4. In order to find the total valence electrons in a if5 (iodine pentafluoride) molecule, first of all you should know the valence electrons present in iodine atom as well as fluorine atom. Put one electron pair in each bond4. Web in the if 5 lewis structure iodine (i) is the least electronegative atom and goes in the center of the. Web thus the lewis structure of any compound can be formed using these simple steps! Web steps of drawing if5 lewis structure. First, determine the total number of valence. Web in the if 5 lewis structure iodine (i) is the least electronegative atom and goes in the center of the lewis structure. In order to find the total valence electrons. Web i quickly take you through how to draw the lewis structure of if5 (iodine pentafluoride). Let’s break down each step in more detail. I also go over hybridization, shape and bond angle. 66k views 10 years ago. Added jun 9, 2014 by webtester in chemistry. Web i quickly take you through how to draw the lewis structure of if5 (iodine pentafluoride). Find more chemistry widgets in wolfram|alpha. Web first, we need to draw the lewis structure of if5. (a) draw an appropriate lewis structure, (b) sketch the molecule with an accurate representation of the geometry, indicating bond angles, (c) name the molecular geometry for each. Draw (on paper) a lewis structure for if_5 and answer the following questions based on your drawing. The number of lone pairs = the number of single bonds = the number of double bonds = 2. Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. Draw lewis structures depicting the. For the compounds (i) if5 and (ii) ch3sh, complete the following: Let us follow a few steps. Draw the molecule by placing atoms on the grid and connecting them with bonds. Draw lewis structures depicting the bonding in simple molecules. Put one electron pair in each bond4. Draw lewis structures depicting the bonding in simple molecules. For the central iodine atom: Put one electron pair in each bond4. Draw lewis structures depicting the bonding in simple molecules. When drawing the structure of an ion, be sure to add/subtract electrons to account for. Web in the if 5 lewis structure iodine (i) is the least electronegative atom and goes in the center of the lewis structure. Web lewis structure for if5. Part a draw an appropriate lewis structure for ifs. Web lewis structure of if5 contains five single bonds between the iodine (i) atom and each fluorine (f) atom. Web chemistry questions and answers. In the lewis structure for if 5 there are a total of 42 valence electrons. #3 indicate formal charges on the atoms, if necessary. Web to properly draw the if 5 lewis structure, follow these steps: For the central iodine atom: Draw (on paper) a lewis structure for if_5 and answer the following questions based on your drawing. Draw lewis structures depicting the bonding in simple molecules. Write lewis symbols for neutral atoms and ions. This gives it ax5e1 shape. 66k views 10 years ago. Thus far, we have discussed the various types of bonds that form between atoms and/or ions. In order to find the total valence electrons in a if5 (iodine pentafluoride) molecule, first of all you should know the valence electrons present in iodine atom as well as fluorine atom.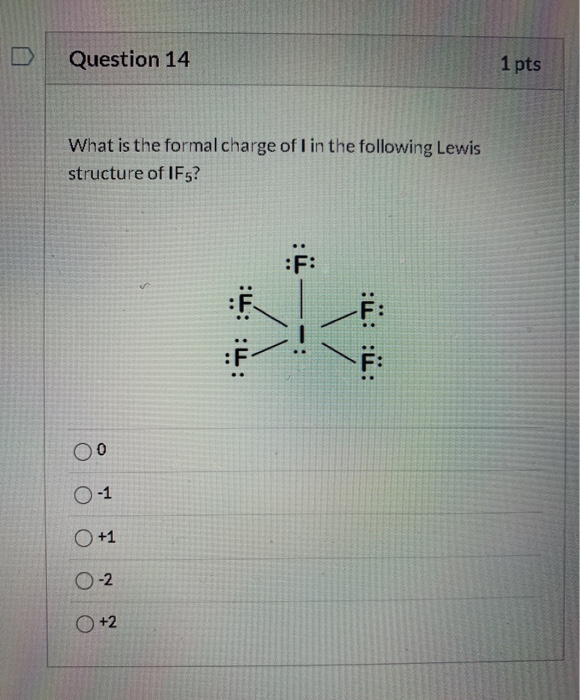
If5 Lewis Structure
IF5 Lewis Structure How to Draw the Lewis Structure for IF5 YouTube
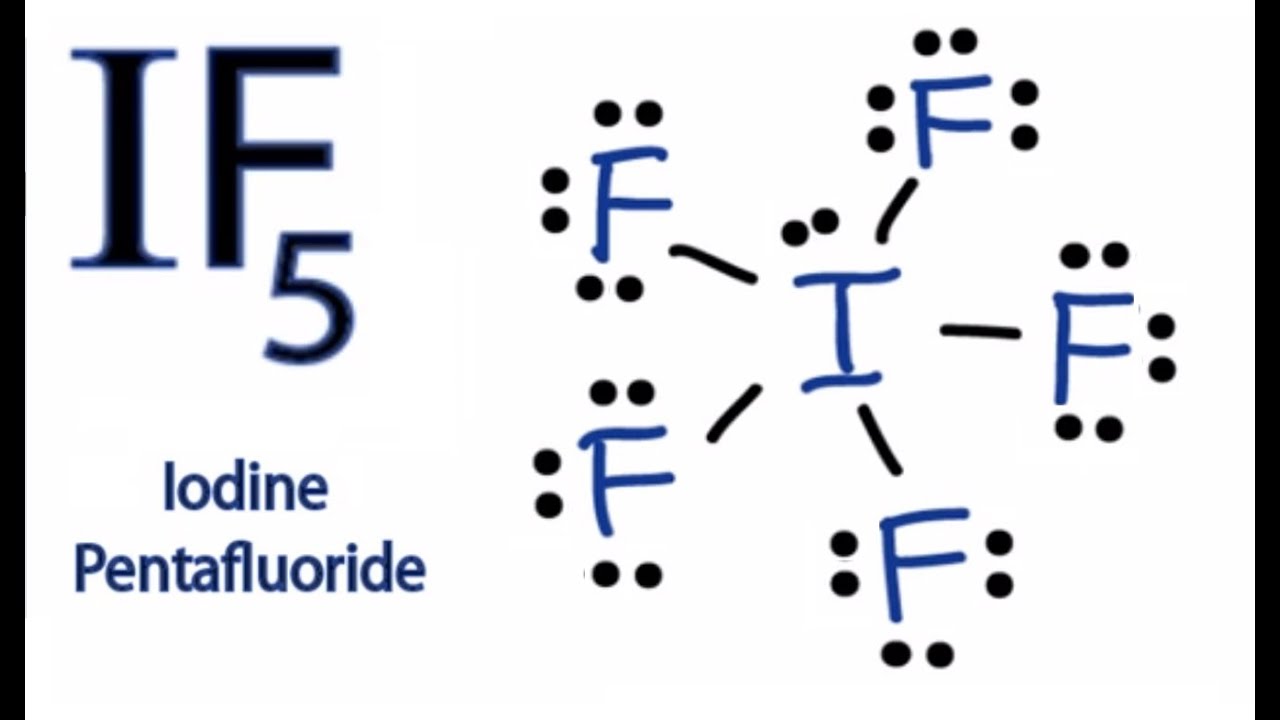
If5 Lewis Structure

So far, we’ve used 42 of the IF5 Lewis structure’s total 42 outermost

How To Draw Lewis Structures A Step By Step Tutorial

IF5 Molecular Geometry, Bond Angles and Electron Geometry YouTube
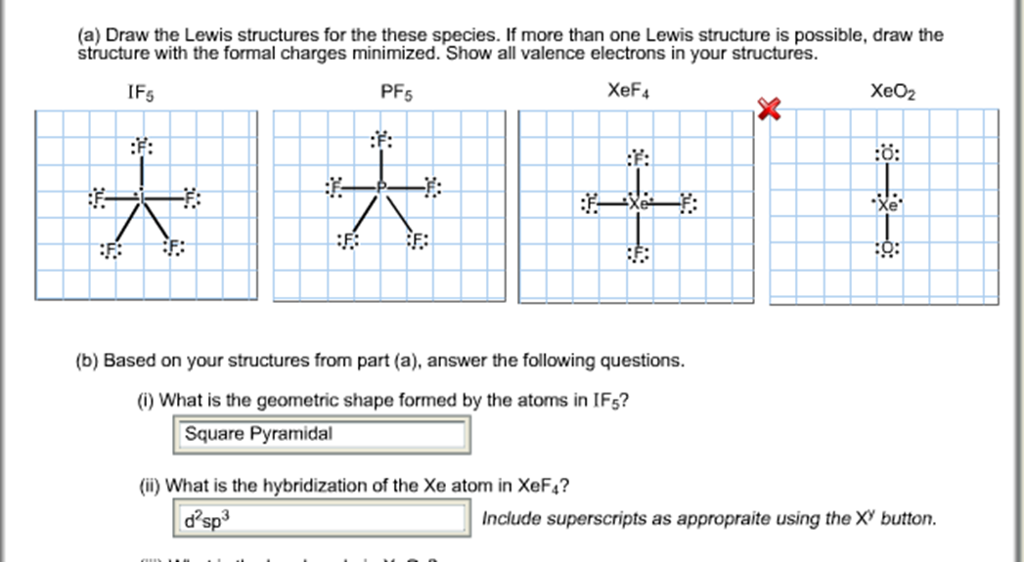
If5 Lewis Structure

IF5 Lewis Structure (Iodine Pentafluoride) YouTube
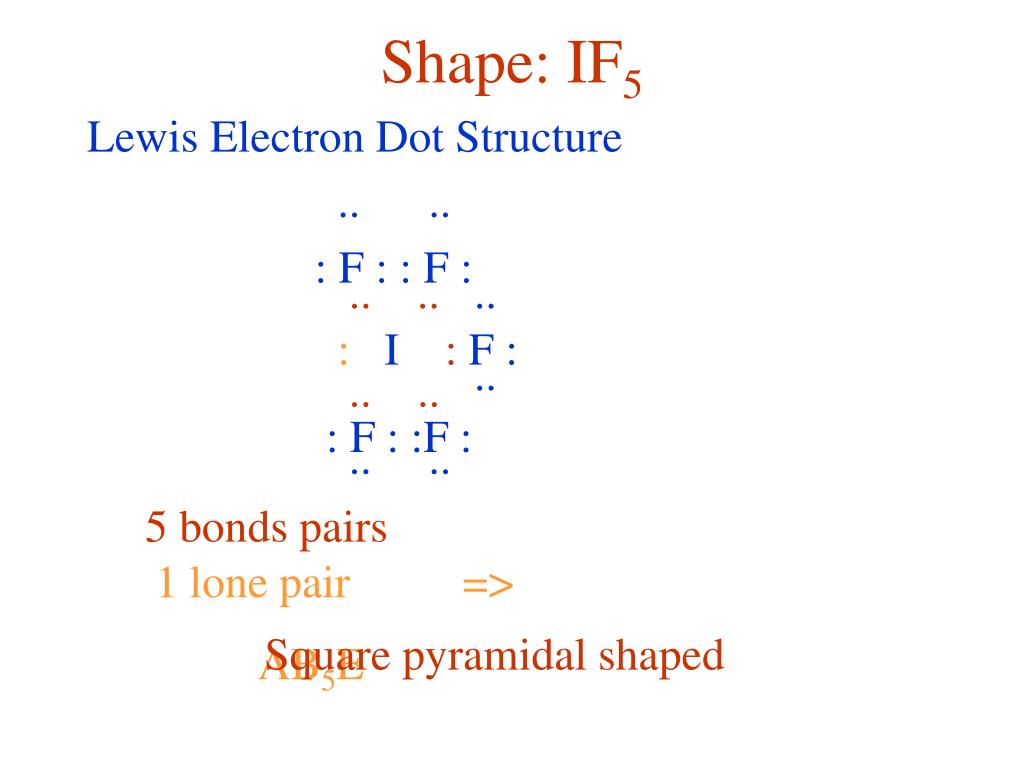
If5 Lewis Structure
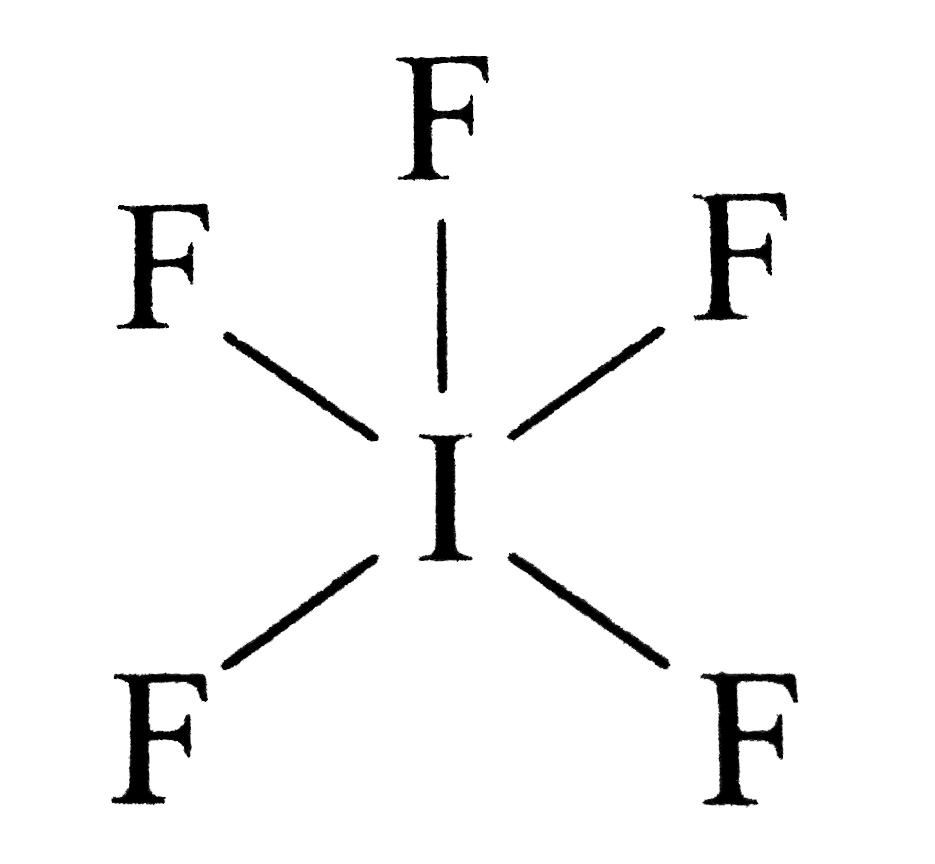
Draw the Lewis structure of iodine pentafluoride, IF5.
(A) Draw An Appropriate Lewis Structure, (B) Sketch The Molecule With An Accurate Representation Of The Geometry, Indicating Bond Angles, (C) Name The Molecular Geometry For Each Central Atom, (D) Determine Whether The Molecule Is Polar, And (E) Identify The Hybridiz.
Web I Quickly Take You Through How To Draw The Lewis Structure Of If5 (Iodine Pentafluoride).
Added Jun 9, 2014 By Webtester In Chemistry.
When Drawing The Structure Of An Ion, Be Sure To Add/Subtract Electrons To Account For.
Related Post: